|
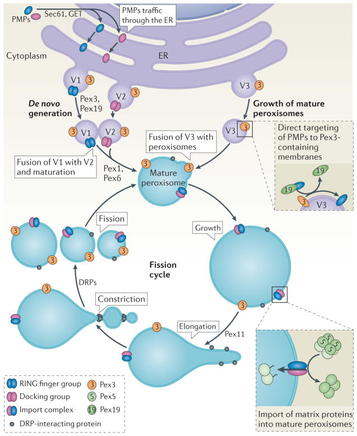
Above is a summary of peroxisome biogenesis, both from the ER and via fission of existing peroxisomes. Click the picture to see a larger image (Smith & Aitchison 2013)
|
Peroxisomes
are created via the actions of a set of at least 32 proteins called
peroxins (Pex). Studies have shown that the peroxisome membrane is
formed firstly via the action of Pex3, followed by Pex19, in association
with the Endoplasmic Reticulum (Kunau 2005),
and it has been found more recently that Pex3 can in fact act on any
endomembrane to synthesise a peroxisome, not just exclusively the ER (Rucktaschel et al 2010). Previously
to these findings, it had been discovered that existing peroxisomes can
take in more proteins then divide via fission to produce new
peroxisomes (Smith & Aitchison 2013).
The proteins required for a peroxisome are translated on free ribosomes in the cytosol, before being imported as complete polypeptide chains into the peroxisome itself. The organelle also requires phospholipids, which are moved to the peroxisome by phospholipid transfer proteins. It is these additions that cause the peroxisome to grow in those that replicate via fission. |
|
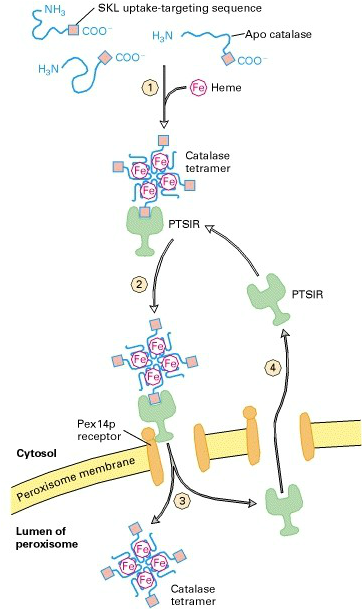
Proteins translocate to the interior of the peroxisome using one of two pathways, which are highly conserved across species. They can have a Ser-Lys-Leu motif at the carboxy terminus, known as peroxisome targeting signal 1 (PTS1), or a nine residue sequence (PTS2). Pex5 codes for PTS1, while PTS2 is coded for by Pex7. The PTS-containing molecules are transported into the peroxisome via translocation complexes, after being recognised by a receptor (Cooper 2000). A major protein that is imported into the peroxisome during its biogenesis is Catalase. The diagram on the right shows the mechanism by which catalase enters the peroxisome. Its PTS is shown, which binds to the PTS1 receptor, which then in turn binds to the membrane bound receptor and facilitates diffusion of the Fe-associated catalase tetramer into the peroxisome (Lodish 2000). |
Image from Lodish 2000
|