|
In the case of 3-methyl branched-chain fatty acids, beta-oxidation is unable to directly take place. This is due to the positioning of the third methyl-group on the fatty acids. Alpha-oxidation is therefore required to remove the last carbon atom to generate 2-methyl fatty acids, which can subsequently be beta-oxidised (Wanders, 2014).
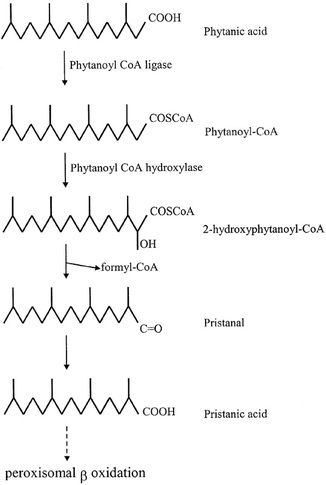
A well known example of such a fatty acid is phytanic acid, which is found in humans. Alpha-oxidation of phytanic acid is shown in diagram to the right. Initially, alpha-oxidation starts with the formation of a CoA-ester called phytanoyl-CoA. This CoA-ester then undergoes hydroxylation, by the enzyme phytanoyl-CoA 2-hydroxylase, to produce 2-hydroxyphytanoyl-CoA. This molecule is then cleaved to form pristanal and formyl-CoA. Formyl-CoA is hydrolysed in a side reaction to produce formic acid and CoA-SH. The chemical compound pristinal is however oxidised to pristianic acid, which after activation to its CoA-ester, pristanoyl-CoA, undergoes beta-oxidation in the peroxisomes. Once beta-oxidised, the end products are transported to the mitochondria for full oxidation to carbon dioxide and water (Wanders 2014). |
Adapted from Wills et al. (2001)
|